CAS NO.: 75-36-5 EINECS No.: 200-865-6
Molecular formula: CH3COCl
Structural Formula:
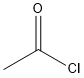
Acetyl chloride is distillated from our own research and developed technique method – consecutive production of Acetyl chloride and HEDP.
At present, our annual production capacity is 8000MTs. In order to keep stable quality and high purity, we use GC equipment to real-time monitor it.
Specification:
| Items |
Index |
|
Appearance |
Clear, colorless fuming liquid |
|
Content(direct injection by GC) |
99.0%
min |
Application:
Acetyl chloride is an important organic synthetic intermediate and acetylation agent, widely used in the field of pesticides, pharmaceuticals, new electroplating complexing and many other fine organic chemicals synthesis. In the pharmaceutical field, it is often used as raw material of acetylsalicylic acid; N-Phenylacetamide; 2,4 – dichloroacetophenone; 4-Phenylacetophenone; Acetazolamide; 2-Acetylfuran; 2-Acetylthiophene; 3-Acetylthianaphthene; 1,1'-Diacetylferrocene, N-Acetylthiourea; 1,4-Naphthalenedicarboxylicacid ; 2-Acetyl-6-methoxynaphthalene; Triphenylmethanol; 4-Ethylbenzoic acid; Acetylcholine chloride; bromadiolone; nabumetone; paracetamol and ibuprofen.
Package:
ISO Tank, 200L Steel-plastic composite barrels and 200L HMHDPE drum or based on customers’ requirement.
Storage:
Avoid contact with water in storage. Keep sealed in a cool, dry, well-ventilated place Acetyl chloride is stable under ordinary conditions but will fume when exposed to atmospheric moisture to form HCl and acetic acid.
Synonyms:
Ethanoyl chloride
Acetic acid chloride
acyl chloride